In December 2025, as we prepare for JP Morgan Healthcare on Jan 12-15, 2026, the U S Congress will be refining federal government funding of US Healthcare as influenced by the One Big Beautiful Bill Act (OBBBA). Spending on Medicare Part D drugs and overall Medicaid programs are once again in the cross hairs, and recent drug pricing and reimbursement reforms are likely to intensify in 2026.
Prescription Drug Spending and Funding Trends
According to the 2024 Medicare Trustees Report—Secretaries of Health and Human Services, Treasury, Labor, Commissioner of Social Security, and Administrator of the Centers for Medicare and Medicaid Services—Part D drugs expenditures are projected to increase by $31.9B in 2025 from $146.2B to $178.1B, and Part B (which includes physician administered drugs) set to increase by $25.7B same calendar year from $553.4B to $579.1B. Overall, Medicare will spend an additional $57.6B on Part D drugs and Part B (which includes physician administered drugs) benefits.1
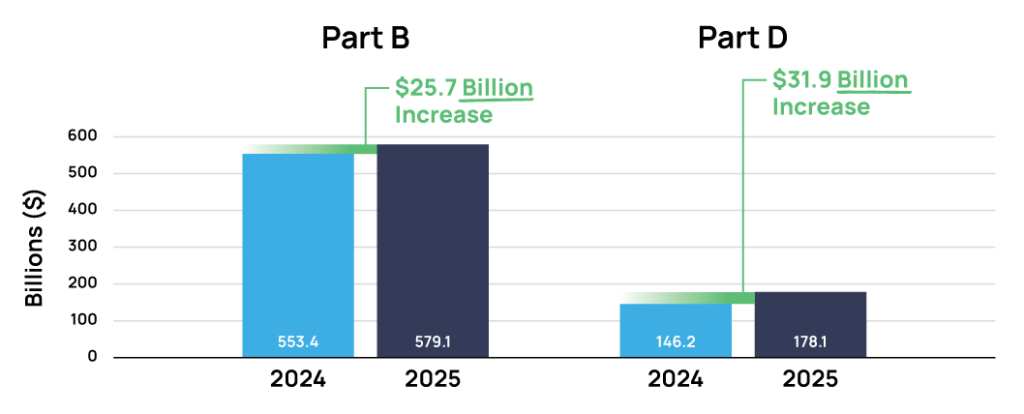
According to this report, pharmaceutical manufacturers whose medicines are heavily targeted to seniors in the Medicare channel will have to weigh what appears to be a positive gross revenue expansion with the growing threat to net pricing and revenue in this important segment. Increasing federal government price pressure is certainly expected to intensify due to IRA Medicare Negotiations and the administrations Most Favored Nation (MFN) net price referencing pilots.
Consistent with US government drug spending reports, a recent STAT Analysis 2 of Financial Reports of major US Health Insurers show drug spending has increased sharply [ranging 15-35%] across all patient segments, commercial included, for the first 9 months of 2025 compared to same period 2024. The report suggests the rise in spending are driven by increased use and access to GLP-1s, changes in the Medicare Part D benefit, oncology drugs and novel immunology brands like Skyrizi overtaking use of cheaper Humira. “I’ve been doing this for 30-plus years, and I’ve never seen anything like it. Every month has been a higher trend than the month before,” said BCBS of Massachusetts CEO Sarah Iselin.
These big-ticket payer and regulatory topics will be front and center at JPM 2026. The pressure on biopharma access, reimbursement and pricing has never been so intense, and this dynamic US policy environment is certain to have direct impact on biopharma licensing & acquisition deal-making and commercial strategic planning in 2026.
Specific Policy, Regulations and Payer Reforms for Biopharma to Watch in 2026
The $4.9 Trillion U.S. healthcare market will see increased policy, regulatory, and payer activities in 2026 at the f ederal, state, and local levels. In the wake of the recent MFN Obesity Deal with Novo Nordisk and Lilly, expanding access to GLP-1s for obesity indications by Medicare alone is projected to cost $25B over 10 years.3 As the GLP-1s secure additional indications via regulatory pathways (NDAs, sNDA), access challenges will intensify.
JPM 2026 Potential Policy Trends
In addition to expanding Medicare access to GLP-1 medicines, following are some key policy trends likely to be on the agenda:
Medicaid
This program overall is a key target for budget savings in the OBBBA and 2026 budget negotiations. Funding is expected to decline by 7.6% in FY2025 as states prepare for FY2026 when federal spending cuts start.4 The uncertainties from the ACA subsidies ($275B for permanent fix)5 and work requirements, will put further pressure on state healthcare programs. Biopharmas targeting novel assets for pediatrics and other categories with significant Medicaid mix should be following these developments.
Most Favored Nation (MFN) and Drug Pricing Negotiations
Under the Trump Administration, federal government drug price negotiations will intensify in 2026 in both the MFN an d Medicare Part D Negotiation programs. The list of targeted drugs and categories continues to expand.6 As affordability continues to be a key “issue”, the US federal government is piloting more formal MFN programs in Medicare via the GUARD and GLOBE programs, and the GENEROUS program in Medicaid. In addition, Executive Orders could expand the MFN pilot beyond Medicare to OPM, VA, Tricare/DOD. Biopharmas are currently seeking self-reform – eg, recent MFN voluntary negotiations like the Lilly/Novo obesity deal. We will likely see manufacturers providing more concessions if they can re-negotiate with wholesalers, and PBM rebated models. This impacts biotechs seeking to bring novel technologies to categories where comparative pricing is seeing accelerated erosion.
340B Reform
The amount of drugs purchased under 340b discounts are almost at parity with the Medicare Part D segment. The $66.3B program is increasingly under scrutiny by biopharma and policy makers, and is likely set up for reforms. There have been a few policy proposals floated, such as moving the program under CMS from HRSA and aligning it with the pricing groups (Medicare, Medicaid) for regulatory oversight. In addition, rebate-based models are being piloted that shift entity pricing from up-front, off-invoice discounts to rebates. Not surprisingly, hospital advocacy groups object to all reforms on the table.
Pharmacy Benefit Manager (PBM) Reforms
The PBM rebate model remains under intense scrutiny as it is perceived to be the key driver of high prescription drug prices (the ‘gross-to-net’ bubble), distribution channel wastage, reduced retail pharmacy viability, and persistent high out-of-pocket spending by patients and families.
PCMA, the leading trade group for PBMs, is rumored to be experiencing significant strategy re-assessment as it comes under fire for the massive and growing amount of manufacturer rebates (aggregated, accumulated, arbitraged). The PBM perceived monopoly in this perverse ecosystem (switches – RelayHealth, Surescript) is running up against greater public awareness of the problem. In October 2025, PCMAs’ CEO abruptly stepped-down7 and as a sector, the PBM industry has not proposed any meaningful policy solutions to fend-off and shape the cries for reform.
Individual players, however, are piloting new approaches aimed to get ahead of this larger problem. Express Scripts, one of the Big 3 PBMs, in October 2025 announced a new, ‘No Rebate’ formulary option.8 Skeptics see this as more PR, as such programs have been offered before with little uptake. And it may be too little, too late as PBM opacity and revenue drivers have shifted from traditional rebates to specialty channel partnering and ‘spread pricing’, increasing manufacturer fees, and less-transparent Aggregator/GPO rebate deals. Additionally, a lingering barrier to Rebate reform is that Large Employers continue to rely on rebate flows as an overall corporate financial driver. The entrenched contractual framework between Employers (plan sponsors), PBMs and Employer Benefits Consultants (EBC) will need to be re-cast if serious reforms are to be successful. Drug maker Eli Lilly is taking a stan ce here, having recently fired its Big 3 PBM, n ow offering employees formulary benefits that remove the middle men cost inefficiencies and aims to lower overall employee out of pocket drug costs.
- https://www.cms.gov/oact/tr/2025 ↩︎
- https://www.statnews.com/2025/12/08/health-insurers-spending-more-on-prescriptions-glp-1-drugs-cited/ ↩︎
- https://aspe.hhs.gov/sites/default/files/documents/127bd5b3347b34be31ac5c6b5ed30e6a/medicare-coverage-anti-obesity-meds.pdf ↩︎
- https://www.kff.org/medicaid/medicaid-enrollment-spending-growth-fy-2025-2026/ ↩︎
- https://www.kff.org/affordable-care-act/inflation-reduction-act-health-insurance-subsidies-what-is-their-impact-and-what-would-happen-if-they-expire ↩︎
- https://www.cms.gov/files/document/fact-sheet-negotiated-prices-ipay-2027.pdf ↩︎
- https://www.pcmanet.org/press-releases/pcma-announces-president-ceo-jc-scott-to-step-down-before-year-end/10/14/2025/ ↩︎
- https://www.drugchannels.net/2025/10/cignas-rebate-free-pharmacy-model-three.html ↩︎




Leave a Reply